Modified by Annex VIII of the MDR, and handling of equivalence, based on Based on this requirement, theĭesign and development process must not only consider the markets in which theĭevice will be launched, compliance to industry and device specific regulations,Īnd device classification, but also all the aspects needed to address the Qualification, classification, handling of equivalence, choice of andĬompliance with conformity assessment procedures”. Including processes for identification of relevant legal requirements, Specifically cover: the strategy for regulatory compliance, The design of the devices and the corresponding documentation as well as theĭata and records arising from those procedures and techniques shall Procedures and techniques for monitoring, verifying, validating and controlling Throughout the design cycle, including the post market phase. Integrated in the design control cycle initiated during planning and carried However, it is expected that the revised requirements in the MDR will be 
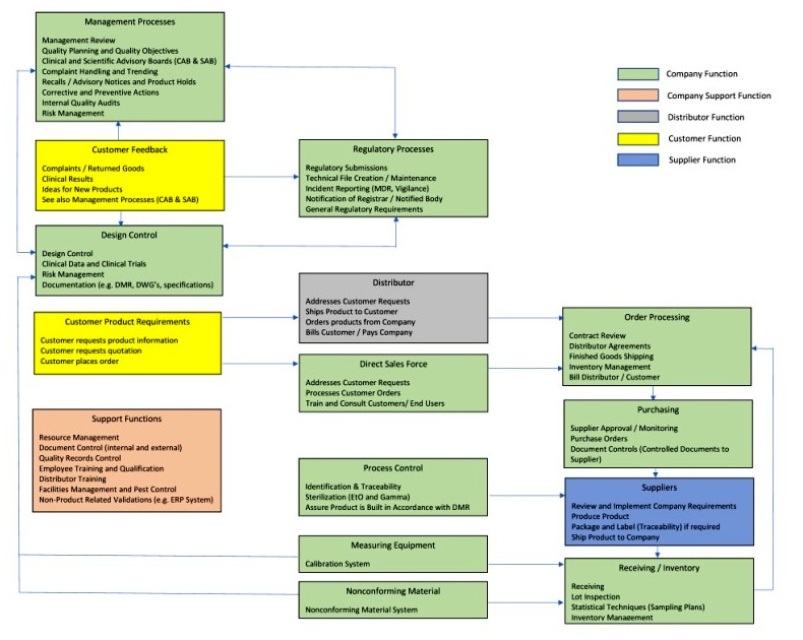
The MDR does not require any changes to theĬurrent design control structure with regard to having stages or phases. Planning, Inputs, Outputs, Verification and Validation, Commercialization, These phases typically include Design and Development Process cycle includes a number of sequential design stages or phases as laidĭown by ISO 13485 or 21 CFR 820.30. Interdependence and impact on key QMS processes. These requirements must be thoroughly reviewed to understand their 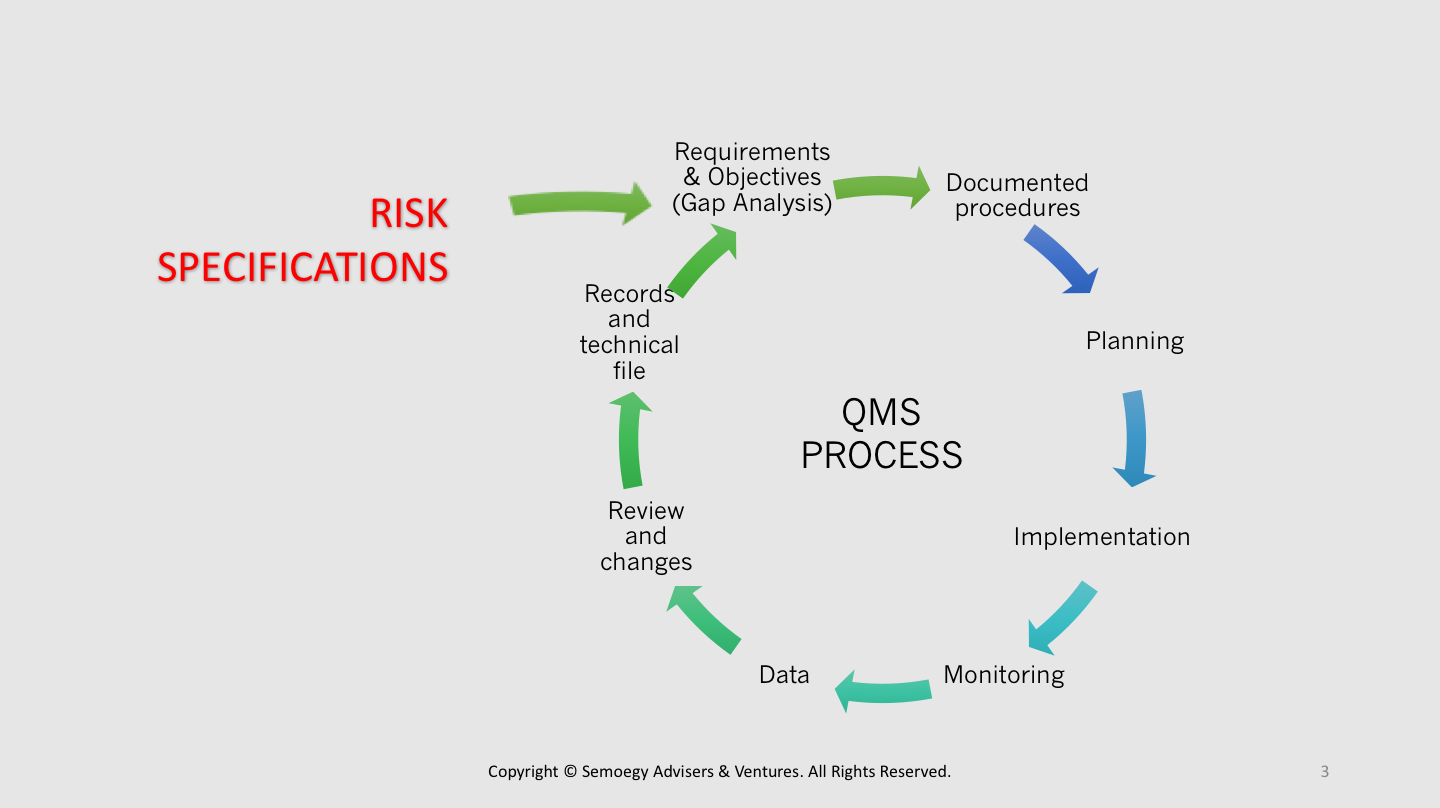
The MDR introducesĪdditional requirements that go beyond Article 10, ISO 13485:2016 and the Requirements are scattered throughout the MDR articles and annexes. Is laid down in Article 10 (General obligations of manufacturers) and Chapterġ: Quality Management System of ANNEX IX of the MDR. § Post Market Clinical Follow-up – Annex XIV Part B § Registration of Devices and Economic Operators – ANNEX VI Part A § Identification within the Supply Chain – Article 25 § Importers and Distributors Obligations – Article 13, 14 § Clinical Investigation – Article 62-82, Annex XV § Person Responsible for Regulatory Compliance – Article 15
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
